1. Introduction
In the textile industry, starting with raw cotton, each process step, including bleaching, dyeing, soaping, softening, and finishing, creates complex chains where dozens of different chemicals are used together. A small imbalance in any link in this chain can lead to serious quality problems, such as permanent stains on the fabric surface, shade variations, stiffness, or dye adhesion.
Polymer-based chemicals (PVA, PVAc, acrylate derivatives, CMC), in particular, are among the auxiliaries widely used in textile processes. These are preferred by businesses to increase yarn strength, prevent wrinkles in finishes, improve post-dyeing handle, or reduce costs. However, these polymers can leave residue on the fabric surface as a result of incorrect dosing, improper process conditions, or insufficient rinsing.
These residues pose serious risks not only to aesthetics but also to the productivity of the business:
-
Dye differences cause customer complaints,
-
Hard handle or gloss differences lead to export refunds,
-
Poor quality product image damages the brand,
-
Unnecessary chemical consumption increases operating costs.
Moreover, this situation not only It is not caused by unintended errors. Sometimes, adding too much polymer to the formulation for cost-cutting purposes or using low-quality chemicals also increases the risk of polymer residue on the fabric. In some cases, fraudulent practices (for example, adding high levels of PVA to increase water retention) can damage both production quality and customer confidence.
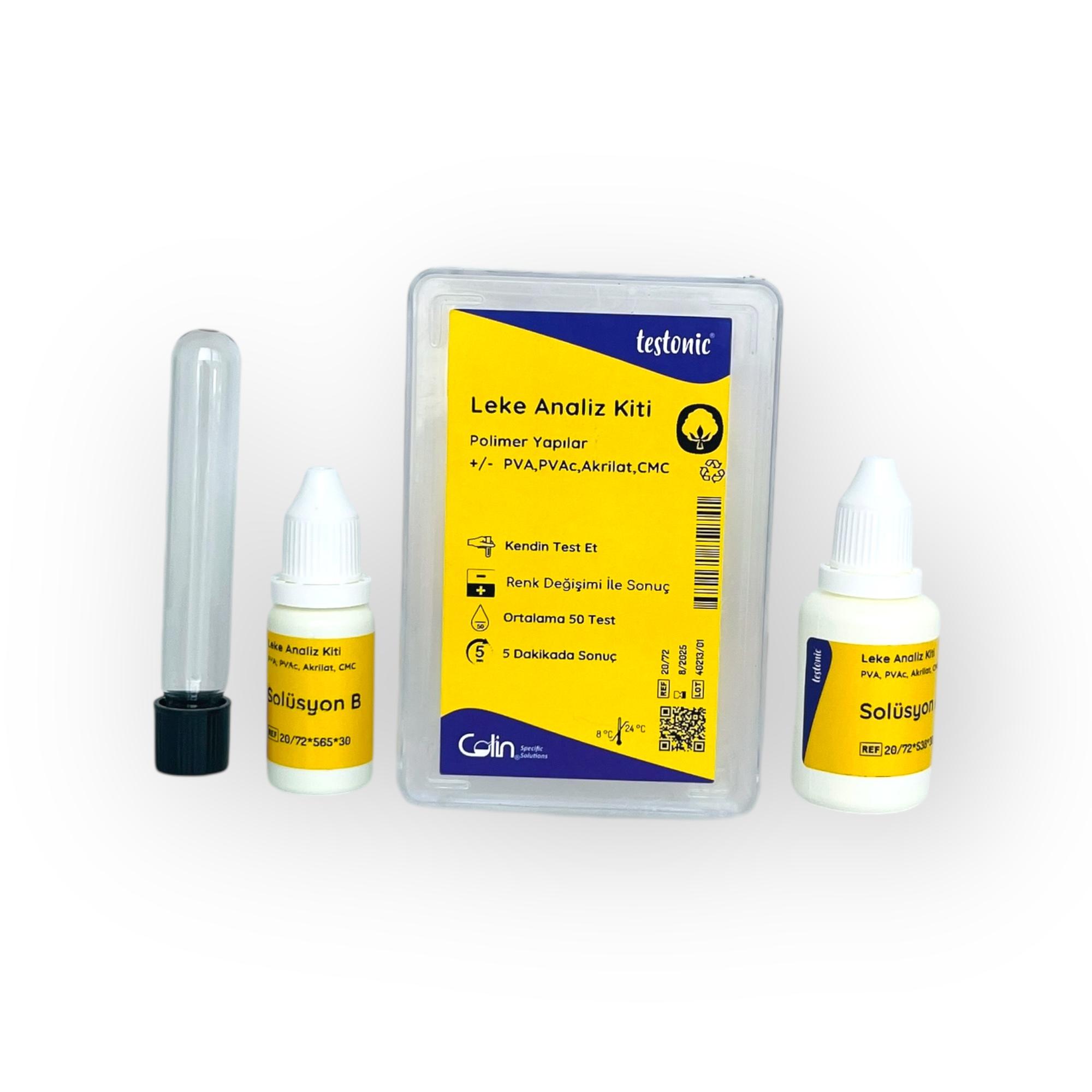
At this point, the Stain Analysis Kit – Polymer Structures (PVA, PVAc, Acrylate, CMC) makes these invisible problems in the production chain visible. The kit allows you to determine in just minutes whether unspecified stains on fabric are truly polymer-related. This rapid and practical analysis provides a significant advantage to quality assurance units, process engineers, and business managers in the decision-making process.
As a result, in modern textile production, controlling polymer-based residues is no longer a choice, but a necessity. The Stain Analysis Kit is a scientific and practical tool that meets this requirement in the field, improving quality assurance and optimizing cost management for businesses.
2. Which Chemicals Contain Polymeric Structures?
Polymeric chemicals used in textile processing are generally characterized by their carrier, binder, film former, or viscosity regulator functions. However, these same properties can cause undesirable cracking, filming, or surface defects under the wrong processing conditions.
The following table summarizes the four main polymer groups:
|
Polymer |
Product Groups Used |
Purpose |
Risk-Creating Conditions |
|---|---|---|---|
|
PVA (Polyvinyl Alcohol) |
Sizing agents, specific strength enhancers, glue additives, some finishing resins |
Increasing yarn strength, increasing abrasion resistance, film formation |
Insufficient desizing & residue on fabric; desizing with Ca²⁺ in hard water; gel formation with borate after alkali; high-temperature adhesive film |
|
PVAc (Polyvinyl Acetate) |
Resin dispersions, adhesives, edge-starching chemicals |
Flexibility, adhesion, wrinkle resistance and finishing effect |
Alkaline saponification during bleaching → conversion to PVA; residue after insufficient rinsing; Film formation during hot drying on MFFT |
|
Acrylate Polymers |
Finishing resins (anti-wrinkle, water-repellent), softener dispersions, binder systems |
Surface smoothness, handle improvement, water-repellency, binding |
Flocking in hard water; high edestabilization in the presence of lectrolytes (with reactive dyeing salts); irregular film on drying close to MFFT. |
|
CMC (Carboxymethyl Cellulose) |
Soaping aids, thickeners in printing pastes, viscosity-regulating additives. |
Viscosity increase, dispersion stabilization, ease of adhesion on the fiber. |
Cross-linking with Ca²⁺/Mg²⁺ in hard water → flocculation; Loss of solubility at low pH; sticky layer when not rinsed. |
2.1. PVA (Polyvinyl Alcohol)
-
Use: Most common as a sizing agent; increases strength by forming a film on the yarn. It is also used in finishing resins for its adhesive properties.
-
Advantage: Reduces yarn breakage before dyeing and provides process stability.
-
Risk: If desizing is inadequate, dye-resistant stains will appear on the fabric after dyeing. In the presence of hard water and borate, PVA forms a gel, leaving a layer that is even more difficult to remove.
2.2. PVAc (Polyvinyl Acetate)
-
Usage: Common in edge starching and finishing; acts as a binder in resin dispersions.
-
Advantage: Protects the fabric's shape and provides crease resistance.
-
Risk: During bleaching, PVAc saponifies in an alkaline environment and transforms into PVA; this new form loses its solubility, leaving a film on the fabric surface. Incorrect drying temperatures make this film permanent.
2.3. Acrylate Polymers
-
Usage: Water-repellent finishes, anti-crease agents, binder dispersions, softener systems.
-
Advantage: Highly effective in improving surface smoothness and handle.
-
Risk: In hard water or high-salt reactive dyeing baths, the dispersion loses its stability and forms flocs. These flocs are observed as spots and cloudiness on the fabric surface. Additionally, incorrect MFFT adjustments can lead to uncontrolled film formation.
2.4. CMC (Carboxymethyl Cellulose)
-
Usage: Thickener in printing pastes, viscosity regulator in soaping aids.
-
Advantage: High viscosity ensures clarity in print patterns; facilitates removal of dyes from the fiber during soaping.
-
Risk: In hard water, it forms flocs by bridging with Ca²⁺/Mg²⁺; It may lose its solubility at low pH. If these flocks adhere to the fabric, they cause a "dull" and "hard" feeling.
3. Conditions for Deposition on Fabric
The following headings will explain the deposition/film-forming mechanisms of PVA, PVAc, acrylate, and CMC residues: I provide relevant process steps (raw cotton/sizing, bleaching, peroxide bleaching, dyeing, washing-soaping, softening, finishing) and examples of combined effects.
3.1. pH and chemical transformations
Mechanism. pH changes determine polymer solubility and loading; they also lead to chemical transformations in some polymers, such as PVAc.
-
PVAc → PVA (alkaline saponification): At high pH (before bleaching), the acetate groups of PVAc hydrolyze, forming PVA; this transformation increases viscosity and the risk of sticky film formation. Thus, while initially PVAc, PVA residue can be detected at the end of the process.
-
CMC (anionic) and acrylate dispersions: At low pH, protonation of carboxylates reduces solubility; As pH decreases, flocculation tendency may increase.
Process examples.
-
Bleaching (NaOH): PVAc saponification accelerates; if proper rinsing is not performed, a PVA film remains. Combined effect: Alkali + hard water = soaps/anionic additives with Ca/Mglar öker also “adheres”PVA to the surface”.
-
Acidic neutralization/special baths: Protonation in CMC → temporary turbidity/shock; if rinsing is not adequate, surface adhesion increases.
3.2. Hard water (Ca²⁺/Mg²⁺) and polyvalent ions
Mechanism. Polyvalent ions such as Ca²⁺/Mg²⁺ weaken the zeta potential of anionic polymers, increasing coagulation/adsorption and the formation of inorganic sediments (limestone, silicate, “lime-soap”).
Process examples and combined effects.
-
Bleaching / washing-soaping: calcium-soaps (lime-soap) and Ca/Mg salts are formed in hard water; These inorganic substances can act as carriers and bind polymers to the fabric. Hard water + alkali = accelerated dissolution.
-
Finish / acrylic binder: Acrylate (most anionic) dispersions tend to flocculate in hard water and in the presence of electrolytes due to charge loss. Hard water + electrolyte (NaCl/Na₂SO₄) = decreased latex stability.
-
Peroxide bleach: Silicate stabilizer + hard water = silicate/calcium salt sediments; fabric “hard handle” and exhibits a dull surface.
3.3. Electrolytes (NaCl, Na₂SO₄) and ionic strength
Mechanism. Electrolytes reduce the stability of colloids such as anionic latex/CMC by compressing the bilayer; flocs/cells form. Electrolytes used in reactive dyeing can increase dye absorption while decreasing the stability of copolymers.
Example of a combined effect.
-
Hard water + high electrolyte (reactive dyeing) = increased risk of acrylic binder/CMC flocculation and spotting/mottled.
3.4. Temperature, drying, and MFFT
Mechanism. Drying acrylic/PVAc emulsion binders above the minimum film formation temperature (MFFT) will yield a continuous film; filming at the wrong place/time → "sticking/gray film" on the fabric. Drying below the MFFT results in a powdery, flaky film and an irregular surface.
Process examples.
-
Finishing/stenter: Excessive thermal load causes the dispersion in the bath to accumulate, leaving a film on the fabric.
-
Post-dyeing drying: Acrylate/PVAc particles may coalesce near the MFFT, forming visible film spots.
3.5. Interaction of oppositely charged auxiliaries (coacervation/coagulation)
Mechanism. The mixing of cationic softeners (quat) and anionic polymer/anionic surfactants forms a complex coacervate or tricel; the resulting complexes provide strong adhesion to the fabric. can be adsorbed (risk of residue during the rinse phase).
Process examples and combined effects.
-
Softening (cationic) + CMC/acrylate/anionic detergent residue: Opposite charge + insufficient rinsing = coacervate film and a shiny surface when rubbed.
-
Soaping residue after dyeing + cationic softener: Sequencing error or insufficient rinsing leaves a thin sticky layer.
3.6. Oxidants, metal catalysts, and silicate stabilizers
Mechanism. Sodium silicate used in peroxide bleaching (H₂O₂/NaOH) can leave silicate precipitates if the appropriate ratio/pH and softening are not applied; in hard water, an inorganic film forms with Ca/Mg, impairing the hold and affecting subsequent dyeing. Switching to organic/non-silicate stabilizers is an industry trend addressing this problem.
Example of a combined effect.
-
Hard water + silicate stabilizer + insufficient rinsing = silicate contamination; acrylate/PVA adhesion is facilitated.
3.7. Dilution/flushing and “dilution-induced” accumulation
Mechanism. During the rinsing/dilution phase of the baths, anionic polymer/surfactant mixtures may shift to coacervation; phase separation may occur with dilution.The solution is diluted and a thin film/spot remains.
Process example.
-
Soaping & rinsing transition: If the dilution rate is high, local coacervate accumulation and adhesion to the surface are observed in the flow regime.
3.8. Special: PVA-Borate complexation (unexpected film/gel transformation)
Mechanism. PVA forms a gel/cross-link via diol-bridging with the borate ion; In the presence of borates (e.g., perborate residues/borate-containing systems), PVA transitions to a gelatinous phase, which can leave a permanent film.
Process example and combined effect.
-
Bleaching/detergent traces (from borate) + sizing residue PVA: Borate + PVA + warm-alkaline medium = rapid gelation/shrinkage, difficult to rinse off.
3.9. Polymer-specific summary (quick checklist)
-
PVA
-
Risky conditions: borate contact after alkali; hard water; insufficient rinsing; hot drying on MFFT.
-
Combined accelerator: Alkali (PVAc→PVA) + borate + hard water → gel/film.
-
-
PVAc
-
Risky conditions: Saponification with Bleaching (NaOH) → Transforms into PVA; now carries the risks of PVA.
-
Combined accelerator: Alkali + high temperature + poor rinsing → adhesive film.
-
-
Acrylate (binder/latex)
-
Risky conditions: Hard water, high electrolyte, MFFT near-superior drying, contact with cationic softener.
-
Combined accelerator: Hard water + NaCl/Na₂SO₄ (reactive dyeing) + drying → flock/film.
-
-
CMC
-
Risky conditions: Complex/cross-linked with Ca²⁺/Mg²⁺; solubility decrease at low pH; high electrolyte.
-
Combined accelerator: Hard water + pH fluctuation (soaping/rinsing) → floc/adsorption.
-
3.10. Practical measures (connect with test kit)
-
Hard water control and chelator use: Ca/Mg chelation (DECOLIN DC in dyeing and washing baths, DECOWET BS in bleaching baths) and silicate-free peroxide stabilizers prevent inorganic sediment from “carrying” polymers. If polymer staining is confirmed with the kit, acrylate sequestrants and stabilizers should be checked first.
-
Sorting and rinsing: After anionic steps, a complete rinse should be performed before cationic softening; otherwise, a coacervate-film remains (a PVA/acrylate/CMC signal may be visible in the kit).
-
MFFT and drying profile: The finish drying-curing temperature must be adjusted according to the MFFT of the binder; otherwise, film formation will start in the "wrong place." The kit provides quick confirmation in the field.
-
Alkali management: In lines using PVAc, excessive alkali/time increases the PVAc→PVA conversion; Particular caution is required if there is a possibility of PVA-borate contact.
-
Dilution regime: Dilute gradually during the soaping and rinsing cycle to prevent microflocks formed by dilution-induced coacervation from adhering to the fabric.
4. Uses and Risks in Textile Processing
4.1. Sizing (Sizing Preparation)
-
Polymers used: PVA, PVAc, partly CMC.
-
Purpose: To increase yarn strength, increase weaving efficiency.
-
Risk: If excess PVA/PVAc is not completely removed during sizing, “dye-proof spots” may occur after dyeing. occurs.
-
Test kit benefits: Quickly reveals whether such stains are actually caused by polymer residue.
4.2. Bleaching (Alkaline Boiling)
-
Polymers used: PVAc (saponifies to PVA), CMC additives.
-
Purpose: To remove natural impurities from the fiber, obtaining a more hydrophilic structure.
-
Risk:
-
PVAc in alkaline medium → PVA transformation, adhesive film layer.
-
CMC flocculation in hard water.
-
-
Test kit benefit: Clarifies whether the residue on the fabric surface is from PVA/CMC.
4.3. Bleaching (Alkaline Peroxide)
-
Polymers used: CMC additives, PVA/PVAc residues carried over from previous processes, acrylate polymers from some finishing chemicals.
-
Purpose: To obtain a brighter, whiter surface by oxidizing the natural colorants in the fiber with hydrogen peroxide and NaOH.
-
Risks (especially acrylate polymers):
-
Acrylate polymers are mostly anionic dispersions. They normally appear stable in alkaline environments.
-
However, when they come into contact with the hard water ions (Ca, Mg) present in the peroxide bath and the calcium salts released from the cotton, ion bridging occurs.
-
This interaction bonds the acrylate chains together and flocculation begins.
-
The flocs remain on the fabric surface as a white, powdery residue.
-
This layer is difficult to remove during rinsing and will not be absorbed by the fabric during subsequent dyeing. This causes the fabric to appear dull, with color tone differences and loss of handle.
-
-
Combined effect:
-
Hard water + high alkali (NaOH) = flocculation develops much faster.
-
Calcium + hard water ions from cotton = increases the amount of sediment.
-
High drying temperature = as the flocks film and adhere to the fabric surface.
-
-
Test kit benefits: It indicates within minutes whether the white layer seen after bleaching is caused by inorganic lime/silicate or actually an acrylate polymer. If a positive result is obtained, the solution for the facility is to review water softening, stabilizer selection, or acrylate additives.
4.4. Dyeing
-
Polymers used: Acrylate dispersions, CMC additives.
-
Purpose: To stabilize dye particles, ensuring homogeneous color distribution.
-
Risk: Hard water and salt loads (NaCl, Na₂SO₄) disrupt the dispersion, causing acrylate/CMC flocculation. Spots and tone differences are visible on the fabric.
-
Test kit benefit: Reveals whether the tone difference is due to polymer residue.
4.5. Soaping
-
Polymers used: CMC, PVA residues.
-
Purpose: To remove dye residues, increase fastness.
-
Risk: After insufficient rinsing, CMC residue leaves a sticky layer on the fabric. When switching to a cationic softener, coacervate is formed through anionic-cationic interactions.
-
Test kit benefit: Confirms the presence of polymer residue in cases of hard handle or dulling problems.
4.6. Softening
-
Polymers used: Acrylate binders, PVAc additives.
-
Purpose: Smooth the fabric surface, provide a soft handle.
-
Risk: Oppositely charged Sticky film due to mixing of chemicals; irregular filming with incorrect MFFT settings.
-
Test kit benefit: Indicates that the stickiness on the fabric after softening is due to the polymer.
4.7. Finishing
-
Polymers used: PVAc, acrylate dispersions, PVA.
-
Purpose: Wrinkle resistance, water repellency, toughness.
-
Risk: Excessive polymer use leaves a hard and sticky layer. Incorrect drying temperature causes film irregularities.
-
Test kit benefits: Determines whether the surface film is polymer-derived.
5. Why is a Test Kit Necessary?
Accurately identifying the source of surface defects in textile production is often difficult. A stain or film appearing on the fabric can be caused by inorganic residue (calcium carbonate, silicate), organic oil/dirt residue, or a polymer-derived film. Misdiagnosis at this point can lead to both the application of incorrect solutions and loss of time and cost.
Stain Analysis Kit – Polymer Structures were developed precisely to eliminate this confusion.
5.1. Correct Diagnosis, Correct Solution
-
Problem: It is often impossible to distinguish visually whether the white layer on the fabric surface is inorganic limescale or acrylate deposits.
-
Solution: Thanks to the kit, the chemical structure of the stains is revealed within minutes. Finding the right source prevents incorrect chemical interventions.
5.2. Cost Management
-
Problem: Excess polymer use often goes unnoticed, but increases costs.
-
Solution: The kit allows businesses to reduce unnecessary polymer consumption by indicating whether the residue is actually caused by polymer.
5.3. Preventing Fraudulent or Substandard Applications
-
Problem: Excessive PVA is added to increase fabric weight, excessive use of substandard PVA, or the addition of cheap acrylate dispersions to the mix.
-
Solution: The kit removes residue from such fraudulent or substandard applications. Thus, quality control units protect the business and the customer.
5.4. Process Improvement and Customer Satisfaction
-
Problem: Errors such as shade differences, rough handling, or a matte appearance often lead to discussions on the production line: is the dye wrong, is the finish wrong, or is the rinsing insufficient?
-
Solution: The kit quickly pinpoints the source of the problem, allowing engineers to focus on the correct process. This reduces customer complaints and prevents export refunds.
5.5. Time and Field Practicality
-
Problem: Sending samples to the laboratory and waiting for detailed analysis slows down production speed.
-
Solution: The kit can be applied in the field within 5 minutes. This speed minimizes time loss, especially in large production lines.
5.6. General Assessment
In brief:
-
For quality assurance, it provides accurate diagnosis.
-
For cost management, it reveals unnecessary polymer usage.
-
It is a security mechanism against fraud.
-
For process optimization, it clarifies the source of the error.
-
For customer satisfaction, it clarifies the source of the error. prevents quality problems.
In conclusion, the Stain Analysis Kit is an indispensable tool in textile production, not just for identifying the problem but for identifying its cause.
6. Conclusion / Closing
Success in textile production means not just producing the fabric, but also for delivering each batch to the customer with the same quality and confidence. Today, polymers like PVA, PVAc, acrylate, and CMC are indispensable for businesses. However, when these same polymers are used incorrectly due to incorrect processing, hard water, insufficient rinsing, or cost pressures, they result in irreversible quality losses, returns, and customer complaints.
The Testonic Stain Analysis Kit was developed to eliminate these risks. It identifies the source of unspecified stains and film on the fabric surface in just a few minutes.Fast, practical, and reliable… Protects your business’s time, costs, and reputation.
On your production line:
-
Predict the risk of paint adhesion,
-
Diagnose hard-to-handle and dull surfaces at their source,
-
Reduce unnecessary polymer consumption,
-
Always offer your customers the same reliable quality.
No more leaving it to chance. Measure and ensure quality in your business with the Testonic Stain Analysis Kit.
Frequently Asked Questions (FAQ)
1. What polymers can the Stain Analysis Kit detect?
PVA, PVAc, acrylate derivatives, and CMC.
2. How does the kit work?
It provides results in minutes with visual detection based on color change.
3. Why is acrylate polymer degradation important?
Alkaline peroxide + hard water ions → Acrylate flocculation & white sediment.
4. What benefits does the kit offer businesses?
Quality assurance, cost control, process improvement, customer satisfaction, and time savings.
5. Can the kit be used in the field?
Yes, it is portable and can be applied without a laboratory.